Evidence From a Systematic Review or Meta-analysis of All Relevant Rcts Semaglutide Prediabetes
Abstruse
Introduction
Orally administered semaglutide is the first glucagon-like peptide one receptor agonist (GLP-1 RA) for oral administration. Every bit head-to-head trials assessing orally administered semaglutide as an addition to 1–2 oral antidiabetic drugs (OADs) vs other GLP-1 RAs are limited, a network meta-assay (NMA) was performed to assess the relative efficacy and safety of orally administered semaglutide 14 mg once-daily (QD) vs injectable GLP-1 RAs in patients with type 2 diabetes inadequately controlled on 1–2 OADs.
Methods
A systematic literature review was conducted to identify randomised controlled trials of GLP-one RAs in patients inadequately controlled on one–two OADs. Information at 26 ± iv weeks were extracted for efficacy and safe outcomes viable for the NMA: change from baseline in glycated haemoglobin (HbA1c), weight, HbA1c target levels (< seven.0% and ≤ 6.v%), blood pressure level, and whatsoever gastrointestinal adverse events specified in system organ class. Information were synthesised using NMA and a Bayesian framework.
Results
In full, 27 studies were included in the analyses. Orally administered semaglutide fourteen mg QD was associated with significantly greater reductions in HbA1c vs near comparators, and numerically greater reductions vs semaglutide 0.v mg in one case-weekly (QW), dulaglutide i.5 mg QW and liraglutide 1.8 mg QD. HbA1c reductions with semaglutide 1 mg QW were numerically greater than those with orally administered semaglutide 14 mg QD. Reductions in trunk weight for orally administered semaglutide 14 mg QD were significantly greater than all comparators except semaglutide QW (both doses). Orally administered semaglutide QD 14 mg was associated with statistically similar odds of experiencing gastrointestinal adverse events vs injectable GLP-i RAs.
Conclusion
Orally administered semaglutide 14 mg QD as an add-on to 1–ii OADs is ane of the nearly efficacious GLP-1 RAs for reducing HbA1c and body weight at 26 ± 4 weeks. Orally administered semaglutide 14 mg QD is well tolerated, with a rubber profile in line with the GLP-1 RA class.
Funding
Novo Nordisk.
Introduction
Type 2 diabetes (T2D) is a chronic and progressive metabolic disorder characterised by elevated levels of blood glucose (hyperglycaemia) [1]. Prolonged, suboptimal glycaemic control is associated with an increased risk of macrovascular (east.g. myocardial infarction, stroke and heart failure) and microvascular (eastward.grand. retinopathy, neuropathy and nephropathy) complications which can reduce life expectancy, adversely impact patients' quality of life and increment the overall treatment costs associated with T2D [2,three,iv,v,6].
The goal for the treatment of T2D is to preclude or delay complications and maintain quality of life, which requires good glycaemic control and management of cardiovascular (CV) risk factors [vii, eight]. Given the progressive nature of the disease, T2D often requires an intensification of handling over fourth dimension to attain glycaemic targets, assessed by measuring glycated haemoglobin (HbA1c) [7, 8]. Current handling guidelines recommend that patients achieve and maintain a target HbA1c level of either < 7% (53 mmol/mol) or of ≤ 6.5% (48 mmol/mol) while minimising the risk of hypoglycaemia [7,8,nine,10,11]. Despite clinical guideline recommendations, up to 50% of patients across Europe and the The states do not encounter glycaemic targets [12, 13]. This is due to several factors including poor adherence to treatment, delay or failure to initiate or intensify therapy (i.east. therapeutic inertia), likewise every bit the suboptimal efficacy and side furnishings of some treatments [xiv, xv]. Torso weight command is also an important element of glycaemic direction strategies, and information technology is estimated that approximately 90% of adults are overweight or obese at the time of T2D diagnosis [sixteen]. Increased body weight is associated with an increased hazard of CV disease, all-cause mortality and reduced quality of life among people with T2D [17,18,19]. Blood pressure, a surrogate marking of CV risk outcomes, is another outcome of interest in the management of T2D. Accordingly, clinical guidelines recommend adequate blood pressure level control to reduce CV gamble [twenty,21,22].
Targeting the incretin organization has go an important therapeutic arroyo for treating T2D [23]. Glucagon-like peptide 1 receptor agonists (GLP-one RAs) are incretin mimetics with proven benefits in terms of improving glycaemic command without increasing the run a risk of hypoglycaemia [24]. GLP-1 RAs provide significant torso weight reduction, and CV risk reduction has also been observed with some GLP-1 RAs in patients with T2D with CV take a chance [24,25,26,27,28]. The American Diabetes Clan and the European Association for the Study of Diabetes recommend GLP-1 RAs as either second- or third-line agents [i.eastward. as an add-on therapy to 1 or two oral antidiabetic drugs (OADs)] in the treatment algorithm [7]. Furthermore, GLP-1 RAs are specifically recommended for patients with T2D who are overweight or obese past the American Clan of Clinical Endocrinologists, the Canadian Diabetes Association and Britain guidelines [21, 29, 30].
Orally administered semaglutide is the offset and only once-daily (QD) oral GLP-1 RA for the treatment of T2D. It has been extensively studied in the Peptide InnOvatioN for Early diabEtes handling (PIONEER) Phase III clinical trial plan [31,32,33,34]. The efficacy and rubber of orally administered semaglutide QD in patients inadequately controlled on 1–2 OADs has been studied in PIONEER 2 [31], PIONEER 3 [32], PIONEER 4 [34] and PIONEER 7 [33]. PIONEER 4 provides evidence for orally administered semaglutide 14 mg vs a GLP-1 RA in patients inadequately controlled on 1–2 OADs, showing significantly greater reductions in HbA1c and body weight with orally administered semaglutide 14 mg compared with liraglutide and placebo (both treatments + metformin ± sodium/glucose cotransporter 2 inhibitor, SGLT2i) [34]. The PIONEER programme also established that orally administered semaglutide QD is well tolerated [31,32,33,34] and that its safety contour is like to that of liraglutide [34].
A broad range of treatment options are currently available to patients with T2D. Thus, agreement the relative clinical benefits of each handling is very important to allow recommendations on their use within a limited budget. While PIONEER 4 provides robust show on the efficacy and prophylactic of orally administered semaglutide QD vs liraglutide [34], evidence from head-to-head trials between orally administered semaglutide and other GLP-1 RAs is limited. Hence, the objective of this study was to conduct a systematic literature review (SLR) and network meta-analysis (NMA) to assess the relative efficacy and safety of orally administered semaglutide xiv mg QD compared with injectable GLP-1 RAs in patients with T2D inadequately controlled on ane–2 OADs.
Methods
Systematic Literature Review
An SLR was performed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [35] to identify randomised controlled trial (RCT) evidence on a wide range of T2D interventions (i.e. all currently used T2D pharmacotherapies) and patient populations. Methodology and results presented herein are specific to studies reporting on the efficacy and safety of orally administered semaglutide and injectable GLP-ane RAs in patients with T2D who are inadequately controlled on 1–2 OADs. Searches of MEDLINE®, Embase and the Cochrane Library were initially performed via Ovid on 5 April 2016 and about recently updated on 2 January 2019 (see Tabular array S1 in supplementary data for further details). Searches of conference proceedings were besides carried out for the European Clan for the Study of Diabetes, the International Society for Pharmacoeconomics and Outcomes Research (ISPOR), the International Diabetes Federation and the American Diabetes Clan Scientific Sessions. Following a written report screening hierarchy for exclusion, all titles and abstracts identified through the literature searches were screened by ii reviewers to assess whether they met the PICOS (population, interventions, comparators, outcomes, study design) selection criteria (supplementary information, Table S2). Once title and abstract screening were completed, any existing discrepancy between the reviewers regarding written report pick was reconciled. The same two reviewers independently screened full-text manufactures for all studies included during the title and abstract screening phase. When a consensus between the two reviewers could not exist reached, a senior reviewer provided arbitration. In addition, data from digital curves were extracted using digital extraction tools. Whatever discrepancies observed between the data extracted by the two analysts were adjudicated past a third analyst. This commodity is based on previously conducted studies and does not contain any studies with homo participants or animals performed by any of the authors.
NMA Methodology
An NMA was performed in accord with guidance from the National Establish for Health and Care Excellence (NICE), ISPOR and the Cochrane Institute [36,37,38,39,40], to assess the relative efficacy of orally administered semaglutide compared with GLP-1 RAs for the treatment of T2D equally an addition to i–two OADs. In the assay, the principal intervention of involvement was orally administered semaglutide 14 mg QD and the primary comparators of interest were all licensed doses of injectable GLP-1 RAs—liraglutide, dulaglutide, exenatide twice-daily (BID), exenatide extended release, lixisenatide and subcutaneously administered semaglutide once-weekly (QW). Albiglutide was withdrawn from the market in 2018 [41] and therefore was not considered a relevant comparator in the NMA. GLP-1 RAs were often taken with other background antidiabetic medications in the trials. To reduce variability between populations across the unlike trials, the definition of the population receiving an add-on to 1–2 OADs was aligned as closely every bit possible to populations in the relevant PIONEER trials of orally administered semaglutide (the principal intervention of interest). The trial population in PIONEER 2 was patients inadequately controlled on metformin, and the trial populations in PIONEER 3, iv and 7 were patients inadequately controlled on one–two OADs (metformin ± sulfonylureas) in PIONEER iii, metformin ± SGLT2i in PIONEER 4 and one–2 OADs (metformin, sulfonylureas, SGLT2i or thiazolidinediones) in PIONEER 7.
Trials assessing a patient population that aligned with PIONEER trials 2, 3, 4 or seven were considered for analysis and consequently, trials which included merely patients inadequately controlled on two OADs were excluded. Similarly, studies which included less than ninety% of patients inadequately controlled on metformin monotherapy, or on i OAD that was not metformin, were excluded from the analysis to reflect standard of care and align with international guidelines [7].
The PIONEER plan used 2 different estimands. The treatment policy estimand evaluated the handling consequence for all randomised patients regardless of trial product discontinuation and use of rescue medication (data analysed using multiple imputation), whereas the trial product estimand evaluated the treatment event for all randomised patients under the assumption that all patients remained on trial product for the entire planned duration of the trial and did non utilise rescue medication (information analysed using a mixed model for repeated measures) [42]. To allow for accurate comparisons with trials reporting data without the employ of rescue medication, the trial production estimand from the PIONEER trials was used for this NMA.
The identified studies were assessed for data on at least i outcome of interest, as well equally their potential to grade a continued network. A feasibility analysis for generating an evidence network for the 20 outcomes of interest was also conducted (supplementary information, Tabular array S2). The NMA was considered feasible for the following efficacy outcomes: change from baseline in HbA1c; proportion of patients achieving HbA1c < 7% (53 mmol/mol) or ≤ half-dozen.5% (48 mmol/mol); changes from baseline in body weight and claret pressure [i.eastward. systolic claret pressure level (SBP) and diastolic claret force per unit area (DBP)], and safety outcomes including the proportion of patients experiencing any gastrointestinal (GI) adverse events (AEs) as specified in organisation organ class.
A normal likelihood, identity link model was used to perform all analyses of continuous outcomes. Where necessary, a shared parameter model was implemented to account for arm-level as well every bit trial-level data reported in the studies. A binomial likelihood (bold a normal distribution) logit link model was used for the analysis of dichotomous outcomes. Both fixed effects and random effects models were run for each outcome, and the near suitable model was chosen on the basis of two criteria: the deviance data and the average posterior residual deviance.
The NMA models were implemented using WinBUGS software (MRC Biostatistics Unit of measurement, Cambridge, UK) [43] and employed a Bayesian framework with the apply of uninformative prior distributions. Three Markov Monte Carlo chains were used, starting from unlike initial values of selected unknown parameters with a burn-in of 50,000 iterations. Convergence for all models was assessed past analysing history and density plots, and Brooks–Gelman–Rubin diagnostic plots [44]. In addition, autocorrelation plots were assessed to detect the presence of autocorrelation in the bondage. Following this, model convergence inferences were fabricated from data obtained by sampling for a farther 10,000 iterations on the three chains.
Median handling differences or odds ratios (ORs) and an associated 95% credible interval (CrI) are presented for the NMA results. For the continuous outcomes HbA1c (%), body weight (kg), SBP and DBP (mmHg), a treatment associated with a greater mean reduction from baseline is favoured. For efficacy dichotomous outcomes, a treatment associated with an increase in the OR (e.g. college odds for achieving a HbA1c level < vii%) is favoured. For GI AEs, a treatment associated with a decrease in the OR is favoured.
In Bayesian statistics, it is considered that differences exist only where the CrI does non include 0.0 for handling differences, or 1.0 for ORs. In some cases, orally administered semaglutide may be associated with a numerical reduction/increase against a comparator; yet, it is assumed that there is no departure betwixt treatments unless the CrI excludes 0.0 (for handling differences), or 1.0 (for ORs).
The median ranks of each treatment are also provided in the supplementary information (Table S14). A handling with a median rank of 1 is considered the best. If ii drugs are both ranked as the second highest, they volition both be given a lower median rank score (i.e. score 3). The surface under the cumulative ranking curve (SUCRA) is also presented in the supplementary data (Table S13). SUCRA values vary between 0% and 100%; a college SUCRA value indicates an increased likelihood that a handling is in the top rank or 1 of the top ranks [45]. This single numeric value can be a helpful simplification of information about the effect of each handling, enabling easier interpretation of the many alternative results that are often calculated within an NMA network.
NMAs judge handling effects by combining evidence from clinical trials. This involves combining direct and indirect measures of effect, the findings of which may not always be aligned with each other. Therefore, it is of import to examine consistency between the two 'sources' of evidence. Hence, where treatment loops were present in the network diagrams, these were statistically evaluated for inconsistency using Bucher's method [39]. Additional informal checks were too performed past comparing the direct study data with the results of the NMA.
This article does not contain any new studies with human or creature subjects performed by any of the authors.
Results
Identified Publications
In total, 108 publications reporting on 71 unique RCTs were included in the SLR; an overall PRISMA flow diagram of the SLR (searches performed between 5 April 2016 and 2 January 2019) is presented in Fig. 1. Private PRISMA flow diagrams (for each search update between 2016 and 2019) and a list of included trials are provided in the supplementary information (Fig. S1 and Table S3, respectively). Of the 71 trials, 47 trials were considered every bit potentially relevant for inclusion in the NMA. The procedure of excluding the remaining 24 studies is detailed in Fig. S2. All 47 trials considered in the NMA formed a connected network, which allowed for the comparing of orally administered semaglutide QD (14 mg dose) with dulaglutide QW (0.75 mg and 1.5 mg doses), liraglutide QD (one.ii mg and one.8 mg doses) exenatide BID (v.0 µg and 10.0 µg doses), exenatide extended release QW (2 mg dose), semaglutide QW (0.5 mg and 1 mg doses) and lixisenatide QD (twenty µg dose). All secondary comparators were removed from the network every bit they did not contribute to any connections in the network, except for placebo, sitagliptin and insulin glargine which were secondary comparators that continued primary comparators of interest. Therefore, four individual treatment arms (i.east. the pioglitazone arm of the Duration-ii trial, the orally administered semaglutide iii mg and 7 mg artillery of the PIONEER three trial and stock-still combination of insulin glargine and lixisenatide arm of LixiLan-O) and seven complete trials [31, 46,47,48,49,50,51] were removed from the networks. Furthermore, PIONEER vii was removed considering of its different trial pattern (assessing flexible dosing for orally administered semaglutide). This resulted in a total of 39 trials remaining in the network [32, 34, 46, 52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,lxx,71,72,73,74,75,76,77,78,79,fourscore,81,82,83,84,85,86,87].
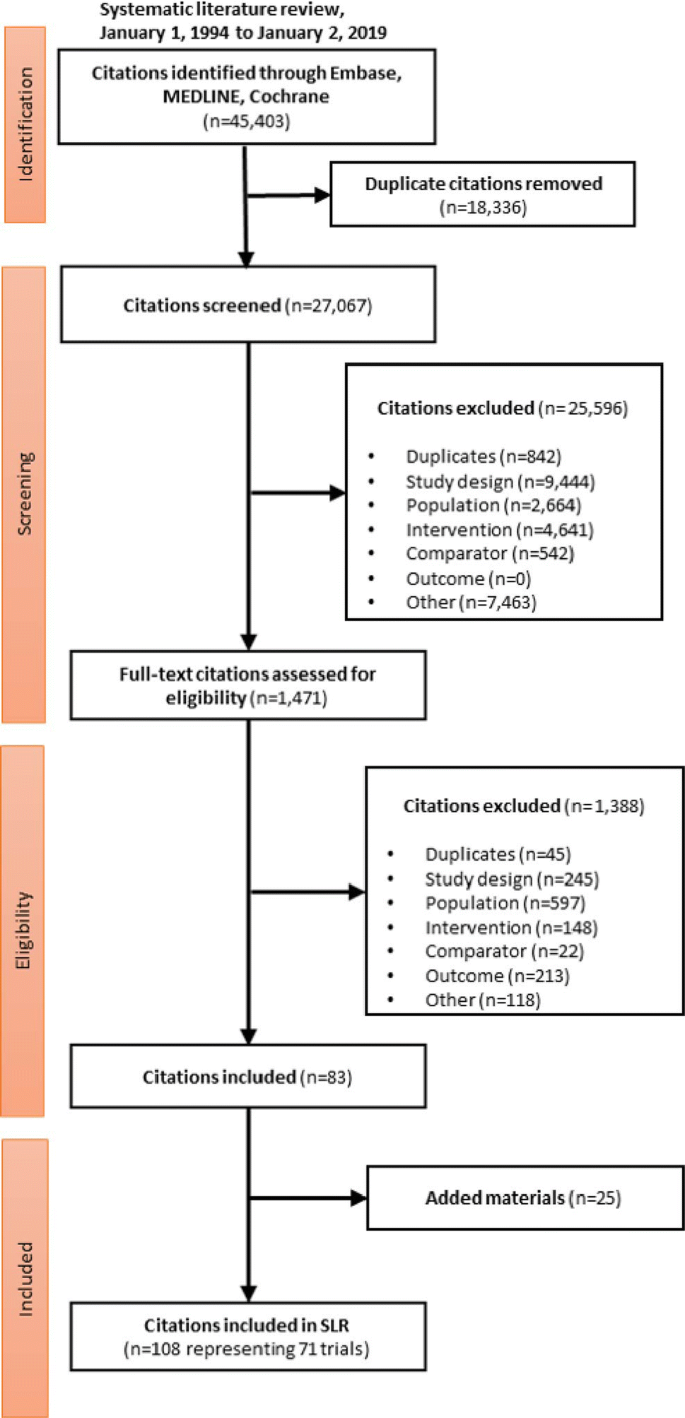
PRISMA flow diagram of the SLR (systematic literature review)
In this network, multiple studies informed the treatment effect for both insulin glargine and lixisenatide. Pooling the insulin glargine arms into a unmarried treatment node was considered clinically appropriate. Similarly, for the lixisenatide treatment artillery, though differences were observed in the titration strategies, these were considered clinically similar such that they could be pooled into a single lixisenatide xx µg QD arm. The only exception to pooling lixisenatide 20 µg doses was with the morning time and evening assistants reported in the GET GOAL-K trial, every bit its master objective was specifically to investigate forenoon vs evening assistants of this handling dose.
The 39 trials retained in the network were examined for time points at which information were available for at least one outcome (supplementary data, Table S4). All 39 trials reported information for at least one effect of interest between 23 and 28 weeks. The level of response to treatment within 4 weeks of the target week was assumed unlikely to vary considerably; therefore, it was considered clinically relevant to analyse each upshot at 26 ± 4 weeks (approximately 6 months). The report pattern and patient characteristics for the 39 trials are presented in the supplementary information (Tabular array S5). Overall, the risk of bias was low across the 39 studies, with the greatest bias run a risk beingness related to elements of unclear or lack of written report blinding and discontinuations.
The majority of trials were considered sufficiently homogenous to be combined in the analysis. Still, eight studies identified in the SLR were considered as potential sources of heterogeneity due to written report design and patient characteristics. Of these eight studies, Derosa 2012 [62] enrolled treatment-naive patients who received metformin for viii months prior to treatment randomisation; Van Gaal 2014 [68] was a study in immature (hateful historic period of 43 years) and obese (hateful body mass index of 36.eight kg/grand2) patients; and six studies (Araki 2015 [70], GET GOAL-1000 Asia [85], Inagaki 2012 [79], Ji 2013 [76], AWARD-CHN2 [81] and Zang 2016 [69]) enrolled only Asian patients. Furthermore, iv trials were excluded from the analyses because of heterogeneity in the groundwork handling and interventions assessed. Duration-NEO-one [82] included patients with 0–3 previous OADs, and relevant subgroup data for the population of involvement for inclusion in the base case NMA were not available. HARMONY-1 [74], HARMONY-3 [46] and HARMONY-4 [75] assessed albiglutide which was withdrawn from the market place in 2018 [41]. It was therefore decided to exclude these 12 studies from the analysis, yielding a total of 27 studies for inclusion in the base example show network [32, 34, 52,53,54,55,56,57,58,59,60,61, 63,64,65,66,67, 71,72,73, 77, 78, 80, 83, 84, 86, 87].
NMA Results
Overall, 27 RCTs reporting outcomes of involvement at 26 ± 4 weeks follow-up were included in the analyses; the evidence network is shown in Fig. 2. In the analysis for the modify from baseline in HbA1c and trunk weight, proportion of patients achieving HbA1c level of < 7% or ≤ 6.5%, and GI AEs, the random furnishings model was chosen as it provided a better fit in terms of deviance information criterion compared with the fixed effects model. In contrast, for the analyses assessing the alter from baseline in SBP and DBP, the fixed effects model was chosen every bit it provided a amend fit than the random furnishings model. The results of the NMA are presented equally handling differences or ORs (orally administered semaglutide QD vs comparator) in Fig. 3. The total matrices of relative treatment effect results are presented in the supplementary information (Tables S6–S12). The associated treatment ranks (SUCRA and median rank) for each result are also presented in the supplementary information (Tables S13 and S14, respectively). In addition, the estimated absolute treatment effects for each result are available in supplementary information (Table S15).
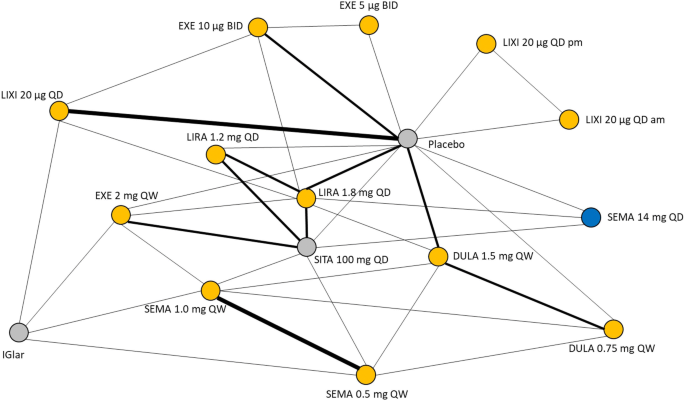
Base of operations case evidence network. Line thickness corresponds to the number of trials contributing to the comparing between two interventions—the thickest equates to three trials, while the thinnest equates to i trial. The blue node indicates the main intervention of interest, orange nodes betoken a primary comparator of interest, and grey nodes indicate a secondary comparator. am morning, BID twice-daily, DULA dulaglutide, EXE exenatide, IGlar insulin glargine, LIRA liraglutide, LIXI lixisenatide, pm evening, QD one time-daily, QW once-weekly, SEMA semaglutide, SITA sitagliptin
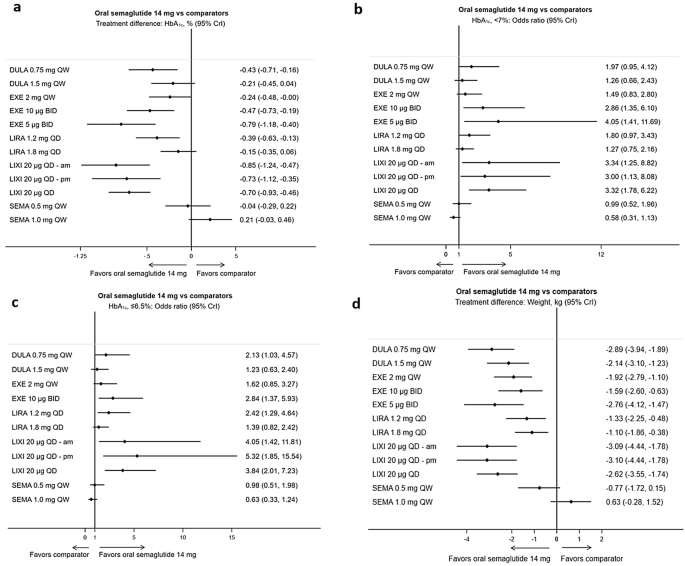
Woods plots of the NMA results: efficacy outcomes for orally administered semaglutide 14 mg QD vs comparators. Treatment differences are considered pregnant when the 95% CrI excludes 0.0. Odds ratios are considered significant when the 95% CrI excludes 1.0. The NMA results are presented as wood plots for a change from baseline in HbA1c, b proportion of patients achieving a HbA1c level < 7% or c HbA1c level ≤ 6.5%, d modify from baseline in body weight. am morn, BID twice-daily, CrI apparent interval, DULA dulaglutide, EXE exenatide, HbA 1c glycated haemoglobin, LIRA liraglutide, LIXI lixisenatide, NMA network meta-analysis, pm evening, QD once-daily, QW once-weekly, SEMA semaglutide
Glycaemic Command
All 27 RCTs reported data on the change from baseline in HbA1c (supplementary information, Table S16); the evidence network for this analysis is shown in Fig. 2. The results (Fig. 3a; Table S6) showed that orally administered semaglutide fourteen mg QD was associated with a significantly greater reduction in HbA1c vs dulaglutide 0.75 mg QW, exenatide BID (both doses), exenatide 2 mg QW, liraglutide 1.two mg QD and lixisenatide QD (all doses). Reductions in HbA1c for orally administered semaglutide 14 mg QD were numerically greater, although non statistically pregnant, compared with semaglutide 0.v mg QW, dulaglutide 1.5 mg QW and liraglutide 1.8 mg QD. Conversely, semaglutide one mg QW was associated with numerically greater HbA1c reductions vs orally administered semaglutide fourteen mg QD. No comparators were identified as significantly better than orally administered semaglutide xiv mg QD.
No significant inconsistency was detected in the network for the change from baseline in HbA1c (p > 0.05), except for one loop of evidence (insulin glargine; exenatide ii mg QW; semaglutide 1 mg QW) for which significant inconsistency (p = 0.006) was detected. When the mean difference in change from baseline betwixt semaglutide 1 mg QW and exenatide 2 mg QW was considered, the issue size direction was the same for both straight and indirect comparisons (directly: − 0.36; 95% confidence interval (CI) − 0.52, − 0.xx; indirect: − 0.74; 95% CI − 0.96, − 0.52); consequently, the NMA too concurred with the direction of effect (− 0.45; 95% CrI − 0.64, − 0.27). While the magnitude of issue differed betwixt the directly, indirect and NMA results, the significant treatment difference favouring semaglutide 1 mg QW over exenatide two mg QW remained in each analysis. Exploratory analyses ended that the touch on on the results of the NMA due to inconsistency in this loop was minimal.
NMAs were also viable for 2 other HbA1c outcomes: HbA1c level < seven% and HbA1c level ≤ half-dozen.5%. The prove networks and data supporting these analyses are shown in the supplementary information (Fig. S3, and Tables S17 and S18, respectively). In line with the results from the assay of the change from baseline in HbA1c, orally administered semaglutide xiv mg QD was associated with significantly higher odds of achieving a HbA1c level < seven% vs exenatide BID (five µg and 10 µg doses) and all dosing schedules of lixisenatide twenty µg (Fig. 3b; Tabular array S7). Orally administered semaglutide xiv mg QD was also associated with significantly college odds of achieving a HbA1c level ≤ 6.v% vs dulaglutide 0.75 mg QW, exenatide 10 µg BID, liraglutide 1.ii mg QD and all dosing schedules of lixisenatide xx µg (Fig. 3c; Table S8). The NMA results likewise suggested that orally administered semaglutide fourteen mg QD was associated with higher odds of achieving target HbA1c levels of < 7% and ≤ 6.five% vs all comparators, except semaglutide QW (both doses), although not all comparisons were statistically pregnant. These analyses bespeak that the improved HbA1c reduction observed with orally administered semaglutide 14 mg QD vs injectable GLP-1 RAs increases the probability of achieving the recommended glycaemic targets.
No inconsistency was detected in most bear witness loops in the networks for the proportion of patients achieving a HbA1c level < vii% or ≤ 6.five%. Still, inconsistency was detected in one loop for HbA1c level < 7% (placebo; sitagliptin 100 mg QD; orally administered semaglutide 14 mg QD), and one loop (placebo; liraglutide 1.8 mg QD; lixisenatide 20 µg QD) for both HbA1c outcomes. As with the change from baseline HbA1c assay, exploratory analyses concluded that there was limited impact on the results of the NMA due to the inconsistency in these loops. Across these analyses, orally administered semaglutide 14 mg QD was one of the highest ranked GLP-1 RAs, achieving SUCRA values of 86–94% (Table S13) and median ranks of ii–three (Tabular array S14). Together, these treatment ranks indicated that orally administered semaglutide fourteen mg QD is the second most clinically efficacious treatment in terms of reduction of HbA1c, and the 3rd virtually effective for achieving a HbA1c level < seven% or ≤ 6.5% at 26 ± 4 weeks, with semaglutide QW being the only treatment ranked college.
Trunk Weight
In total, 27 trials reported the modify from baseline in body weight (supplementary information, Table S19) and the evidence network is shown in Fig. S3. The results showed that orally administered semaglutide 14 mg QD was associated with a significantly greater reduction in trunk weight vs all GLP-1 RA comparators except semaglutide QW (0.v mg and 1 mg doses) (Fig. 3d; Table S9). No significant inconsistency was detected in this network (p > 0.05). Together, the SUCRA values and median ranks betoken that orally administered semaglutide 14 mg QD is the second nigh efficacious GLP-1 RA in terms of body weight reduction at 26 ± 4 weeks (Tabular array S13 and S14).
Blood Pressure
Claret pressure level was assessed in terms of changes from baseline in SBP and DBP. In total, 21 trials reported the change from baseline in SBP, and 20 trials reported the change from baseline in DBP (supplementary information, Tables S20 and S21, respectively). The evidence network for both outcomes is shown in Fig. S3. The analysis suggested that orally administered semaglutide 14 mg QD was associated with a like reduction in SBP vs all GLP-1 RA comparators, except semaglutide 1 mg QW (supplementary information, Table S10). Orally administered semaglutide 14 mg QD was also associated with a like reduction in DBP vs all GLP-i RA comparators (supplementary data, Table S11). No significant inconsistency was detected in the loops of evidence for both networks. The ranking and SUCRA values for blood pressure outcomes are reported in supplementary data (Tables S13 and S14, respectively).
Safety Outcomes
I of the most commonly cited reasons for treatment discontinuation with GLP-1 RAs is GI AEs [88, 89]. Therefore, an analysis of the incidence of the virtually commonly reported GI AEs (e.g. nausea, airsickness, diarrhoea) was performed in this NMA. In total, 17 trials reported the proportion of patients experiencing GI AEs (supplementary information, Tabular array S22) and the evidence network is shown in Fig. S3. The analysis indicated that orally administered semaglutide 14 mg QD was associated with statistically like odds of experiencing GI AEs compared with injectable GLP-1 RAs (supplementary information, Table S12). No pregnant inconsistency was detected in the evidence network for GI AEs (p > 0.05), except in one loop of evidence for which the direction of the effect was the same and there was some overlap of 95% CrIs.
Discussion
The objective of this study was to assess the relative efficacy and safety of orally administered semaglutide xiv mg QD compared with injectable GLP-1 RAs in patients with T2D inadequately controlled on 1–2 OADs. The analyses showed that orally administered semaglutide 14 mg QD was associated with a significantly greater reduction in HbA1c at 26 ± 4 weeks vs about GLP-i RA comparators, and with numerically greater reductions vs semaglutide 0.5 mg QW, dulaglutide 1.5 mg QW and liraglutide i.8 mg QD. Conversely, semaglutide i mg QW was associated with numerically greater HbA1c reductions compared with orally administered semaglutide xiv mg QD. In one case-daily orally administered semaglutide likewise provided a significantly greater reduction in body weight vs all GLP-1 RA comparators except semaglutide QW (0.5 mg and one mg doses) and was associated with similar reductions in SBP and DBP vs all GLP-one RA comparators, except semaglutide i mg QW. Results also suggest that the improved HbA1c reduction observed with orally administered semaglutide xiv mg QD vs injectable GLP-1 RAs, except semaglutide QW (both doses), increases the probability of achieving the recommended glycaemic target levels of < vii% or ≤ half dozen.5%, although not all comparisons were statistically pregnant. The findings from these analyses are supported by treatment ranks and SUCRA values which together indicate that orally administered semaglutide 14 mg QD is the second best handling, after semaglutide 1 mg QW, for reducing HbA1c levels and trunk weight at 26 ± 4 weeks.
A good balance between improvements in efficacy and the chance of AEs is of import for constructive treatment. Gastrointestinal AEs are commonly cited reasons for discontinuing treatment with GLP-1 RAs [88, 89]. Our analysis institute that orally administered semaglutide 14 mg was associated with similar odds of experiencing GI AEs, including nausea, vomiting and diarrhoea, when compared with injectable GLP-1 RAs. This is in line with the findings from the PIONEER clinical trial programme which demonstrated that orally administered semaglutide is well tolerated [31,32,33,34] and has a condom profile similar to that of liraglutide [34]. Inconsistency was detected in i loop of the show network for GI AEs. Along with the subjectivity associated with reporting of GI AEs that may impact the relative treatment effect, potential effect modifiers for outcomes were identified including diet, eating patterns and use of medication to ameliorate GI issues. However, every bit these parameters were not widely reported beyond studies in the network, a meta-regression to examine the impact of these factors on the outcome was not feasible. Furthermore, this loop consisted of only a unmarried trial to inform each handling pair and it was not possible to identify which trial was "deviant" amongst these, thus causing the inconsistency.
To our knowledge, this is the kickoff study assessing the relative efficacy and condom of orally administered semaglutide xiv mg every bit an improver therapy to 1–2 OADs compared with currently available GLP-one RAs. Four recent NMAs assessed the relative efficacy and safe of GLP-1 RAs (not including orally administered semaglutide xiv mg) [90,91,92,93] of which one considered patients with T2D inadequately controlled on 1–ii OADs as its target population [93]. The findings from our NMA in terms of GLP-1 RAs' relative efficacy and ranking are consistent with those reported in the NMA by Witkowski et al. [93].
Findings from this NMA are robust on the basis of the number and homogeneity of trials included in the evidence network, equally well equally the alignment of its methodology with clinical practice guidance from Prissy, ISPOR and the Cochrane Found [36,37,38,39,40]. Furthermore, all the trials included in the NMA were identified in an SLR, ensuring that all available testify was captured. The network also considered RCTs assessing GLP-1 RAs as an improver to 1–two OADs to include all the relevant PIONEER trials that assessed orally administered semaglutide as an improver to OADs. Importantly, the NMA focused on patients with T2D previously receiving 1–two OADs who are a relevant segment of patients routinely receiving GLP-RAs.
The NMA was field of study to common limitations, including some heterogeneity in populations beyond the different trials, the time points reported across the trials, publication bias in the studies included in the SLR, and inconsistency in some testify networks. To minimise variability between trial populations, the definition of population receiving an add together-on to 1–ii OADs was closely aligned to populations in the relevant trials of orally administered semaglutide; therefore, just trials assessing a patient population that aligned with PIONEER trials 2, 3, 4 or seven were considered in the analysis. The heterogeneity in the reported time points was addressed by using a common and well-accustomed approach of time signal window [91, 92, 94]. Overall, the chance of bias was considered depression beyond the studies. While the bulk of trials in the analyses were blinded, 20 trials were open label, which could potentially increment the gamble of operation bias [95]. Inconsistency was also detected in some of the networks in the analyses at 26 ± 4 weeks. Notwithstanding, removing prove to reduce inconsistency is not always considered all-time do [39]. The NMA estimates are a compromise between direct and indirect prove and therefore removing direct evidence for ane handling comparison (which is likewise used as indirect evidence in other treatment comparisons) means that the NMA results are probable to vary and information technology would exist impossible to identify which indirect evidence is non consistent with the direct evidence. Therefore, the presence of inconsistency is a limitation of the NMA results for outcomes where inconsistency was detected.
Conclusion
Orally administered semaglutide 14 mg QD as an add-on to 1–two OADs is one of the almost efficacious GLP-1 RAs for reducing HbA1c levels and body weight, and for achieving target HbA1c levels at 26 ± 4 weeks. In improver, the similar odds of experiencing GI AEs in comparing with other GLP-1 RAs indicate that orally administered semaglutide is well tolerated and non associated with additional burden to patients.
References
-
Forbes JM, Cooper ME. Mechanisms of diabetic complications. Physiol Rev. 2013;93:137–88.
-
Caro JJ, Ward AJ, O'Brien JA. Lifetime costs of complications resulting from blazon two diabetes in the U.S. Diabetes Care. 2002;25:476–81.
-
Diabetes UK. Facts and stats 2016. https://world wide web.diabetes.org.britain/Documents/Position%20statements/DiabetesUK_Facts_Stats_Oct16.pdf. Accessed 14 Aug 2019.
-
Hex North, Bartlett C, Wright D, Taylor M, Varley D. Estimating the current and futurity costs of type i and type 2 diabetes in the Britain, including direct health costs and indirect societal and productivity costs. Diabet Med. 2012;29:855–62.
-
Stratton IM, Adler AI, Neil HA, et al. Clan of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405–12.
-
Williams R, Van Gaal L, Lucioni C. Assessing the touch of complications on the costs of type II diabetes. Diabetologia. 2002;45:S13–7.
-
Davies MJ, D'Alessio DA, Fradkin J, et al. Management of hyperglycemia in type two diabetes, 2018. A consensus report past the American Diabetes Clan (ADA) and the European Association for the Report of Diabetes (EASD). Diabetes Care. 2018;2018(41):2669–701.
-
American Diabetes Association. Standards of medical care in diabetes. Diabetes Intendance. 2019;42:S61–70.
-
Handelsman Y, Bloomgarden ZT, Grunberger One thousand, et al. American Association of Clinical Endocrinologists and American College of Endocrinology—clinical practice guidelines for developing a diabetes mellitus comprehensive intendance plan-2015-executive summary. Endocr Pract. 2015;21:413–37.
-
International Diabetes Federation (IDF). Clinical do recommendations for managing type ii diabetes in primary intendance 2017. https://www.idf.org/e-library/guidelines/128-idf-clinical-practice-recommendations-for-managing-type-two-diabetes-in-main-care.html. Accessed fourteen Aug 2019.
-
American Association of Clinical Endocrinologists and American College of Endocrinology. AACE/ACE comprehensive blazon 2 diabetes direction algorithm; 2019. https://world wide web.aace.com/pdfs/diabetes/AACE_2019_Diabetes_Algorithm_FINAL_ES.pdf. Accessed 14 Aug 2019.
-
Carls G, Huynh J, Tuttle Due east, Yee J, Edelman SV. Achievement of glycated hemoglobin goals in the U.s. remains unchanged through 2014. Diabetes Ther. 2017;eight:863–73.
-
de Pablos-Velasco P, Parhofer KG, Bradley C, et al. Current level of glycaemic command and its associated factors in patients with type 2 diabetes across Europe: data from the PANORAMA study. Clin Endocrinol (Oxf). 2014;80:47–56.
-
Khan H, Lasker SS, Chowdhury TA. Exploring reasons for very poor glycaemic control in patients with type 2 diabetes. Prim Care Diabetes. 2011;5:251–5.
-
Reach G, Pechtner V, Gentilella R, Corcos A, Ceriello A. Clinical inertia and its impact on treatment intensification in people with blazon 2 diabetes mellitus. Diabetes Metab. 2017;43:501–11.
-
Carnethon MR, De Chavez PJ, Biggs ML, et al. Association of weight status with mortality in adults with incident diabetes. JAMA. 2012;308:581–90.
-
Bodegard J, Sundstrom J, Svennblad B, Ostgren CJ, Nilsson PM, Johansson G. Changes in body mass index following newly diagnosed type ii diabetes and risk of cardiovascular mortality: a cohort report of 8486 primary-care patients. Diabetes Metab. 2013;39:306–13.
-
Coffey JT, Brandle M, Zhou H, et al. Valuing health-related quality of life in diabetes. Diabetes Care. 2002;25:2238–43.
-
Oldridge NB, Stump TE, Nothwehr FK, Clark DO. Prevalence and outcomes of comorbid metabolic and cardiovascular conditions in middle- and older-age adults. J Clin Epidemiol. 2001;54:928–34.
-
Authors/Chore Forcefulness Members, Ryden L, Grant PJ, et al. ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: The Job Forcefulness on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur Eye J. 2013;34:3035–87.
-
Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive blazon 2 diabetes direction algorithm—2019 Executive Summary. Endocr Pract. 2019;25:69–100.
-
Lorber D. Importance of cardiovascular disease risk direction in patients with type two diabetes mellitus. Diabetes Metab Syndr Obes. 2014;7:169–83.
-
Garber AJ. Long-acting glucagon-like peptide 1 receptor agonists: a review of their efficacy and tolerability. Diabetes Intendance. 2011;34(Suppl 2):S279–84.
-
Prasad-Reddy L, Isaacs D. A clinical review of GLP-1 receptor agonists: efficacy and rubber in diabetes and beyond. Drugs Context. 2015;4:212283.
-
Marso SP, Daniels GH, Brownish-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type ii diabetes. North Engl J Med. 2016;375:311–22.
-
Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with blazon 2 diabetes. N Engl J Med. 2016;375:1834–44.
-
Isaacs D, Prasad-Reddy L, Srivastava SB. Role of glucagon-like peptide 1 receptor agonists in direction of obesity. Am J Wellness Syst Pharm. 2016;73:1493–507.
-
Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394:121–30.
-
Scottish Intercollegiate Guidelines Network. SIGN 154—a national clinical guideline: pharmacological direction of glycaemic control in people with type ii diabetes; 2017. http://www.sign.ac.uk/assets/sign154.pdf. Accessed 14 Aug 2019.
-
Canadian Diabetes Association Clinical Practice Guidelines Good Committee. Pharmacologic management of type 2 diabetes: Nov 2016 interim update. Can J Diabetes. 2016;40:193–5.
-
Novo Nordisk. Data on file. PIONEER 2—oral semaglutide vs. SGLT-2 Inhibitor CTR, version one.0 03 October 2018. A 52-calendar week randomised, open-label, agile-controlled trial to investigate the efficacy and safety of oral semaglutide versus empagliflozin in subjects with blazon ii diabetes mellitus; 2018.
-
Rosenstock J, Allison D, Birkenfeld AL, et al. Consequence of additional oral semaglutide vs sitagliptin on glycated hemoglobin in adults with type two diabetes uncontrolled with metformin lonely or with sulfonylurea: the PIONEER iii randomized clinical trial. JAMA. 2019;321:1466–80.
-
Pieber TR, Bode B, Mertens A, et al. Efficacy and safety of oral semaglutide with flexible dose adjustment versus sitagliptin in type two diabetes (PIONEER 7): a multicentre, open-label, randomised, stage 3a trial. Lancet Diabetes Endocrinol. 2019;7:528–39.
-
Pratley R, Amod A, Hoff ST, et al. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomised, double-bullheaded, phase 3a trial. Lancet. 2019;394:39–l.
-
Moher D, Liberati A, Tetzlaff J, Altman DG, Prisma Grouping. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535.
-
Dias S, Welton NJ, Sutton AJ, Ades AE. NICE DSU Technical Support Document 2: a generalised linear modelling framework for pairwise and network meta-assay of randomised controlled trials; 2011 [updated April 2014]. http://www.nicedsu.org.britain/TSD2%20General%20meta%20analysis%20corrected%2015April2014.pdf Accessed xiv Aug 2019.
-
Hoaglin DC, Hawkins N, Jansen JP, et al. Conducting indirect-treatment-comparison and network-meta-analysis studies: study of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: role 2. Value Health. 2011;14:429–37.
-
Higgins J, Dark-green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.2; 2009 [updated September 2009]. www.cochrane-handbook.org. Accessed 14 Aug 2019.
-
Dias South, Welton NJ, Sutton AJ, Caldwell DM, Lu Thou, Ades AE. Overnice DSU Technical Support Document 4: inconsistency in networks of evidence based on randomised controlled trials; 2011 [updated Apr 2014]. http://nicedsu.org.britain/wp-content/uploads/2016/03/TSD4-Inconsistency.final_.15April2014.pdf. Accessed 14 Aug 2019.
-
Dias S, Welton NJ, Sutton AJ, Valdwell DM, Guobing L, Ades AE. NICE DSU Technical Support Document three: heterogeneity: subgroups, meta-regression, bias and bias-adjustment; 2011. http://world wide web.nicedsu.org.uk. Accessed xiv Aug 2019.
-
European Medicines Agency. Albiglutide withdrawal. https://world wide web.ema.europa.eu/en/documents/assessment-report/public-argument-eperzan-withdrawal-marketing-authorization-european-union_en.pdf. Accessed Jun 2019.
-
Aroda VR, Saugstrup T, Buse JB, Donsmark G, Zacho J, Davies MJ. Incorporating and interpreting regulatory guidance on estimands in diabetes clinical trials: the PIONEER 1 randomized clinical trial as an example. Diabetes Obes Metab. 2019;21:2203–10.
-
Lunn DJ, Best N, Thomas A, Spiegelhalter D. The BUGS book: a practical introduction to Bayesian analysis. Boca Raton: CRC; 2013.
-
Welton NJ, Sutton AJ, Cooper NJ, Abrams KR, Ades AE. Evidence synthesis for decision making in healthcare. 1st ed. New York: Wiley; 2012.
-
Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-assay: an overview and tutorial. J Clin Epidemiol. 2011;64:163–71.
-
Ahren B, Johnson SL, Stewart M, et al. HARMONY three: 104-week randomized, double-blind, placebo- and active-controlled trial assessing the efficacy and safety of albiglutide compared with placebo, sitagliptin, and glimepiride in patients with type 2 diabetes taking metformin. Diabetes Care. 2014;37:2141–8.
-
Frias JP, Guja C, Hardy E, et al. Exenatide in one case weekly plus dapagliflozin once daily versus exenatide or dapagliflozin lonely in patients with blazon ii diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol. 2016;four:1004–16.
-
Derosa K, Maffioli P, Salvadeo SA, et al. Exenatide versus glibenclamide in patients with diabetes. Diabetes Technol Ther. 2010;12:233–40.
-
Gallwitz B, Guzman J, Dotta F, et al. Exenatide twice daily versus glimepiride for prevention of glycaemic deterioration in patients with blazon 2 diabetes with metformin failure (EUREXA): an open up-characterization, randomised controlled trial. Lancet. 2012;379:2270–8.
-
Derosa G, Putignano P, Bossi Ac, et al. Exenatide or glimepiride added to metformin on metabolic control and on insulin resistance in type 2 diabetic patients. Eur J Pharmacol. 2011;666:251–half-dozen.
-
Gallwitz B, Bohmer M, Segiet T, et al. Exenatide twice daily versus premixed insulin aspart 70/30 in metformin-treated patients with type 2 diabetes: a randomized 26-calendar week report on glycemic command and hypoglycemia. Diabetes Care. 2011;34:604–six.
-
Zinman B, Gerich J, Buse JB, et al. Efficacy and prophylactic of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 Met + TZD). Diabetes Care. 2009;32:1224–30.
-
Buse JB, Rosenstock J, Sesti G, et al. Liraglutide once a solar day versus exenatide twice a 24-hour interval for type 2 diabetes: a 26-week randomised, parallel-grouping, multinational, open-characterization trial (Atomic number 82-half dozen). Lancet. 2009;374:39–47.
-
Pratley RE, Nauck One thousand, Bailey T, et al. Liraglutide versus sitagliptin for patients with type 2 diabetes who did not accept acceptable glycaemic control with metformin: a 26-week, randomised, parallel-group, open up-label trial. Lancet. 2010;375:1447–56.
-
Buse JB, Nauck M, Forst T, et al. Exenatide once weekly versus liraglutide once daily in patients with type 2 diabetes (DURATION-6): a randomised, open-label report. Lancet. 2013;381:117–24.
-
Charbonnel B, Steinberg H, Eymard E, et al. Efficacy and safety over 26 weeks of an oral treatment strategy including sitagliptin compared with an injectable treatment strategy with liraglutide in patients with blazon 2 diabetes mellitus inadequately controlled on metformin: a randomised clinical trial. Diabetologia. 2013;56:1503–xi.
-
Bailey TS, Takacs R, Tinahones FJ, et al. Efficacy and condom of switching from sitagliptin to liraglutide in subjects with blazon two diabetes (LIRA-SWITCH): a randomized, double-blind, double-dummy, active-controlled 26-week trial. Diabetes Obes Metab. 2016;18:1191–8.
-
Ahren B, Masmiquel 50, Kumar H, et al. Efficacy and rubber of in one case-weekly semaglutide versus once-daily sitagliptin equally an add together-on to metformin, thiazolidinediones, or both, in patients with type 2 diabetes (SUSTAIN two): a 56-week, double-blind, phase 3a, randomised trial. Lancet Diabetes Endocrinol. 2017;5:341–54.
-
Aroda VR, Bain SC, Cariou B, et al. Efficacy and condom of one time-weekly semaglutide versus once-daily insulin glargine as add-on to metformin (with or without sulfonylureas) in insulin-naive patients with type 2 diabetes (SUSTAIN 4): a randomised, open-label, parallel-group, multicentre, multinational, phase 3a trial. Lancet Diabetes Endocrinol. 2017;5:355–66.
-
Dungan KM, Povedano ST, Forst T, et al. Once-weekly dulaglutide versus once-daily liraglutide in metformin-treated patients with type 2 diabetes (AWARD-6): a randomised, open-label, phase 3, non-inferiority trial. Lancet. 2014;384:1349–57.
-
Nauck M, Rizzo Grand, Johnson A, Bosch-Traberg H, Madsen J, Cariou B. One time-daily liraglutide versus lixisenatide every bit improver to metformin in type 2 diabetes: a 26-week randomized controlled clinical trial. Diabetes Care. 2016;39:1501–ix.
-
Derosa K, Franzetti IG, Querci F, et al. Exenatide plus metformin compared with metformin alone on beta-jail cell function in patients with type 2 diabetes. Diabet Med. 2012;29:1515–23.
-
Bergenstal RM, Wysham C, Macconell Fifty, et al. Efficacy and rubber of exenatide in one case weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of blazon ii diabetes (DURATION-2): a randomised trial. Lancet. 2010;376:431–9.
-
DeFronzo RA, Ratner RE, Han J, Kim DD, Fineman MS, Baron AD. Effects of exenatide (exendin-iv) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care. 2005;28:1092–100.
-
Bolli GB, Munteanu Chiliad, Dotsenko South, et al. Efficacy and safety of lixisenatide once daily vs. placebo in people with type 2 diabetes insufficiently controlled on metformin (GetGoal-F1). Diabet Med. 2014;31:176–84.
-
Ahren B, Leguizamo Dimas A, Miossec P, Saubadu South, Aronson R. Efficacy and safety of lixisenatide one time-daily morning or evening injections in type 2 diabetes inadequately controlled on metformin (GetGoal-M). Diabetes Care. 2013;36:2543–50.
-
Rosenstock J, Raccah D, Koranyi L, et al. Efficacy and prophylactic of lixisenatide once daily versus exenatide twice daily in type 2 diabetes inadequately controlled on metformin: a 24-week, randomized, open-label, agile-controlled report (GetGoal-X). Diabetes Care. 2013;36:2945–51.
-
Van Gaal 50, Souhami E, Zhou T, Aronson R. Efficacy and safety of the glucagon-like peptide-1 receptor agonist lixisenatide versus the dipeptidyl peptidase-4 inhibitor sitagliptin in young (< fifty years) obese patients with type 2 diabetes mellitus. J Clin Transl Endocrinol. 2014;1:31–7.
-
Zang 50, Liu Y, Geng J, et al. Efficacy and safety of liraglutide versus sitagliptin, both in combination with metformin, in Chinese patients with type 2 diabetes: a 26-week, open up-label, randomized, active comparator clinical trial. Diabetes Obes Metab. 2016;18:803–11.
-
Araki E, Inagaki N, Tanizawa Y, Oura T, Takeuchi M, Imaoka T. Efficacy and safety of once-weekly dulaglutide in combination with sulphonylurea and/or biguanide compared with once-daily insulin glargine in Japanese patients with type 2 diabetes: a randomized, open-characterization, phase Three, not-inferiority study. Diabetes Obes Metab. 2015;17:994–1002.
-
Diamant M, Van Gaal L, Guerci B, et al. Exenatide once weekly versus insulin glargine for type ii diabetes (Elapsing-3): 3-year results of an open-characterization randomised trial. Lancet Diabetes Endocrinol. 2014;2:464–73.
-
Pinget M, Goldenberg R, Niemoeller E, Muehlen-Bartmer I, Guo H, Aronson R. Efficacy and rubber of lixisenatide once daily versus placebo in type ii diabetes insufficiently controlled on pioglitazone (GetGoal-P). Diabetes Obes Metab. 2013;15:k–seven.
-
Rosenstock J, Hanefeld K, Shamanna P, et al. Benign furnishings of once-daily lixisenatide on overall and postprandial glycemic levels without significant excess of hypoglycemia in blazon 2 diabetes inadequately controlled on a sulfonylurea with or without metformin (GetGoal-S). J Diabetes Complicat. 2014;28:386–92.
-
Reusch J, Stewart MW, Perkins CM, et al. Efficacy and prophylactic of once-weekly glucagon-like peptide i receptor agonist albiglutide (HARMONY 1 trial): 52-week primary endpoint results from a randomized, double-blind, placebo-controlled trial in patients with type 2 diabetes mellitus not controlled on pioglitazone, with or without metformin. Diabetes Obes Metab. 2014;16:1257–64.
-
Weissman PN, Carr MC, Ye J, et al. HARMONY four: randomised clinical trial comparing in one case-weekly albiglutide and insulin glargine in patients with type ii diabetes inadequately controlled with metformin with or without sulfonylurea. Diabetologia. 2014;57:2475–84.
-
Ji L, Onishi Y, Ahn CW, et al. Efficacy and safety of exenatide once-weekly vs exenatide twice-daily in Asian patients with type 2 diabetes mellitus. J Diabetes Investig. 2013;4:53–61.
-
Liutkus J, Rosas Guzman J, Norwood P, et al. A placebo-controlled trial of exenatide twice-daily added to thiazolidinediones alone or in combination with metformin. Diabetes Obes Metab. 2010;12:1058–65.
-
Rosenstock J, Aronson R, Grunberger Chiliad, et al. Benefits of LixiLan, a titratable stock-still-ratio combination of insulin glargine plus lixisenatide, versus insulin glargine and lixisenatide monocomponents in blazon two diabetes inadequately controlled on oral agents: the LixiLan-O randomized trial. Diabetes Care. 2016;39:2026–35.
-
Inagaki N, Atsumi Y, Oura T, Saito H, Imaoka T. Efficacy and safety profile of exenatide one time weekly compared with insulin in one case daily in Japanese patients with type 2 diabetes treated with oral antidiabetes drug(due south): results from a 26-week, randomized, open up-label, parallel-group, multicenter, noninferiority written report. Clin Ther. 2012;34(1892–908):e1.
-
Ludvik B, Frias JP, Tinahones FJ, et al. Dulaglutide equally add-on therapy to SGLT2 inhibitors in patients with inadequately controlled type 2 diabetes (AWARD-ten): a 24-week, randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2018;six:370–81.
-
Wang Due west, Nevarez L, Filippova East, et al. Efficacy and safe of once-weekly dulaglutide versus insulin glargine in mainly Asian patients with type 2 diabetes mellitus on metformin and/or a sulphonylurea: a 52-week open up-label, randomized stage Three trial. Diabetes Obes Metab. 2019;21:234–43.
-
Wysham CH, Rosenstock J, Vetter ML, Dong F, Ohman P, Iqbal North. Efficacy and tolerability of the new autoinjected intermission of exenatide one time weekly versus exenatide twice daily in patients with blazon ii diabetes. Diabetes Obes Metab. 2018;20:165–72.
-
Gadde KM, Vetter ML, Iqbal N, Hardy E, Ohman P. Efficacy and safety of autoinjected exenatide once-weekly suspension versus sitagliptin or placebo with metformin in patients with type ii diabetes: the Duration-NEO-two randomized clinical report. Diabetes Obes Metab. 2017;19:979–88.
-
Frias JP, Nauck MA, Van J, et al. Efficacy and safe of LY3298176, a novel dual GIP and GLP-ane receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase ii trial. Lancet. 2018;392:2180–93.
-
Yu Pan C, Han P, Liu X, et al. Lixisenatide treatment improves glycaemic control in Asian patients with type 2 diabetes mellitus inadequately controlled on metformin with or without sulfonylurea: a randomized, double-blind, placebo-controlled, 24-week trial (GetGoal-Grand-Asia). Diabetes Metab Res Rev. 2014;30:726–35.
-
Ahmann AJ, Capehorn M, Charpentier Thou, et al. Efficacy and condom of once-weekly semaglutide versus exenatide ER in subjects with type 2 diabetes (SUSTAIN iii): a 56-week, open up-label, randomized clinical trial. Diabetes Care. 2018;41:258–66.
-
Novo Nordisk. Data on file: results meeting—semaglutide subcutaneous vs. dulaglutide (SUSTAIN seven). 2017.
-
Sun F, Chai S, Yu K, et al. Gastrointestinal adverse events of glucagon-like peptide-1 receptor agonists in patients with type two diabetes: a systematic review and network meta-assay. Diabetes Technol Ther. 2015;17:35–42.
-
Sikirica MV, Martin AA, Wood R, Leith A, Piercy J, Higgins Five. Reasons for discontinuation of GLP1 receptor agonists: data from a real-world cross-exclusive survey of physicians and their patients with type ii diabetes. Diabetes Metab Syndr Obes. 2017;10:403–12.
-
Zaccardi F, Htike ZZ, Webb DR, Khunti M, Davies MJ. Benefits and harms of one time-weekly glucagon-like peptide-1 receptor agonist treatments: a systematic review and network meta-assay. Ann Intern Med. 2016;164:102–13.
-
Kayaniyil S, Lozano-Ortega Chiliad, Bennett HA, et al. A network meta-analysis comparison exenatide in one case weekly with other GLP-ane receptor agonists for the handling of type 2 diabetes mellitus. Diabetes Ther. 2016;7:27–43.
-
Orme ME, Nguyen H, Lu JY, Thomas SA. Comparative effectiveness of glycemic control in patients with blazon 2 diabetes treated with GLP-1 receptor agonists: a network meta-assay of placebo-controlled and active-comparator trials. Diabetes Metab Syndr Obes. 2017;10:111–22.
-
Witkowski Grand, Wilkinson L, Webb N, Weids A, Glah D, Vrazic H. A systematic literature review and network meta-analysis comparing once-weekly semaglutide with other GLP-1 receptor agonists in patients with blazon two diabetes previously receiving one-two oral anti-diabetic drugs. Diabetes Ther. 2018;ix:1149–67.
-
Barnett AH, Orme ME, Fenici P, Townsend R, Wygant G, Roudaut M. Systematic review and network meta-assay to compare dapagliflozin with other diabetes medications in combination with metformin for adults with type two diabetes. Diabetes Intern Med. 2014;S6:006.
-
Turner RM, Spiegelhalter DJ, Smith GC, Thompson SG. Bias modelling in prove synthesis. J R Stat Soc Ser A Stat Soc. 2009;172:21–47.
Acknowledgements
Funding
Sponsorship for this study and the Rapid Service Fee were funded by Novo Nordisk.
Medical Writing, Editorial and Other Aid
Medical writing and editorial aid in the preparation of this manuscript was provided by Sonia Alesso of DRG Abacus; support for this help was funded by Novo Nordisk. The SLR was conducted by DRG Abacus. For the NMA, data analysis was performed Abby Paine of Zedediah Consulting and feasibility cess was conducted by Jatin Gupta of DRG Abacus. SLR and NMA were both funded by Novo Nordisk. The authors would like to give thanks Srikanth Deenadayalan for reviewing the manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the piece of work, and have given terminal blessing to the version to be published.
Disclosures
Solomon Nuhoho is an employee of Novo Nordisk. Brian Bekker Hansen is an employee and shareholder of Novo Nordisk. Tam Dang-Tan is an employee and shareholder of Novo Nordisk. Jatin Gupta is an employee of DRG Abacus. Mary Fletcher-Louis is an employee of DRG Abacus. Abby Paine is the possessor of Zedediah Consulting.
Compliance with Ethics Guidelines
This commodity is based on previously conducted studies and does not contain whatsoever studies with human participants or animals performed by any of the authors.
Data Availability
All information generated or analysed during this study are included in this published article as supplementary data files.
Open Access
This article is distributed nether the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in whatever medium, provided yous requite appropriate credit to the original author(south) and the source, provide a link to the Artistic Commons license, and point if changes were made.
Writer information
Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article become to https://doi.org/10.6084/m9.figshare.9901934.
Electronic Supplementary Fabric
Rights and permissions
This commodity is published under an open access license. Delight check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended utilise exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
Almost this commodity
Cite this article
Nuhoho, S., Gupta, J., Hansen, B.B. et al. Orally Administered Semaglutide Versus GLP-ane RAs in Patients with Type 2 Diabetes Previously Receiving ane–2 Oral Antidiabetics: Systematic Review and Network Meta-Assay. Diabetes Ther ten, 2183–2199 (2019). https://doi.org/10.1007/s13300-019-00706-y
-
Received:
-
Published:
-
Outcome Engagement:
-
DOI : https://doi.org/10.1007/s13300-019-00706-y
Keywords
- Torso weight
- GLP-1 receptor agonist
- Glycaemic control
- HbA1c
- Network meta-analysis
- Orally administered semaglutide
- Systematic literature review
- Type 2 diabetes
Source: https://link.springer.com/article/10.1007/s13300-019-00706-y
0 Response to "Evidence From a Systematic Review or Meta-analysis of All Relevant Rcts Semaglutide Prediabetes"
Post a Comment